Aids in preventing adverse drug reactions
Adverse drug reactions related to pharmacodynamic drug interactions are a significant clinical problem associated with polypharmacy. Polypharmacy is common in older patients and the younger at-risk population (1,2).
Current drug interaction warning systems typically focus on individual drug-drug pairs. Implementing alerts for every conceivable interaction would result in excessive over-alerting within electronic health record (EHR) systems (3). Over-alerting can overwhelm healthcare providers, which can lead to alert fatigue, and undermine the effectiveness of the warning system (4). Medication reviews may reveal potential pharmacodynamic interactions (5).
“Pharmacodynamic drug interactions are a significant clinical problem associated with polypharmacy.”
Clinical solution
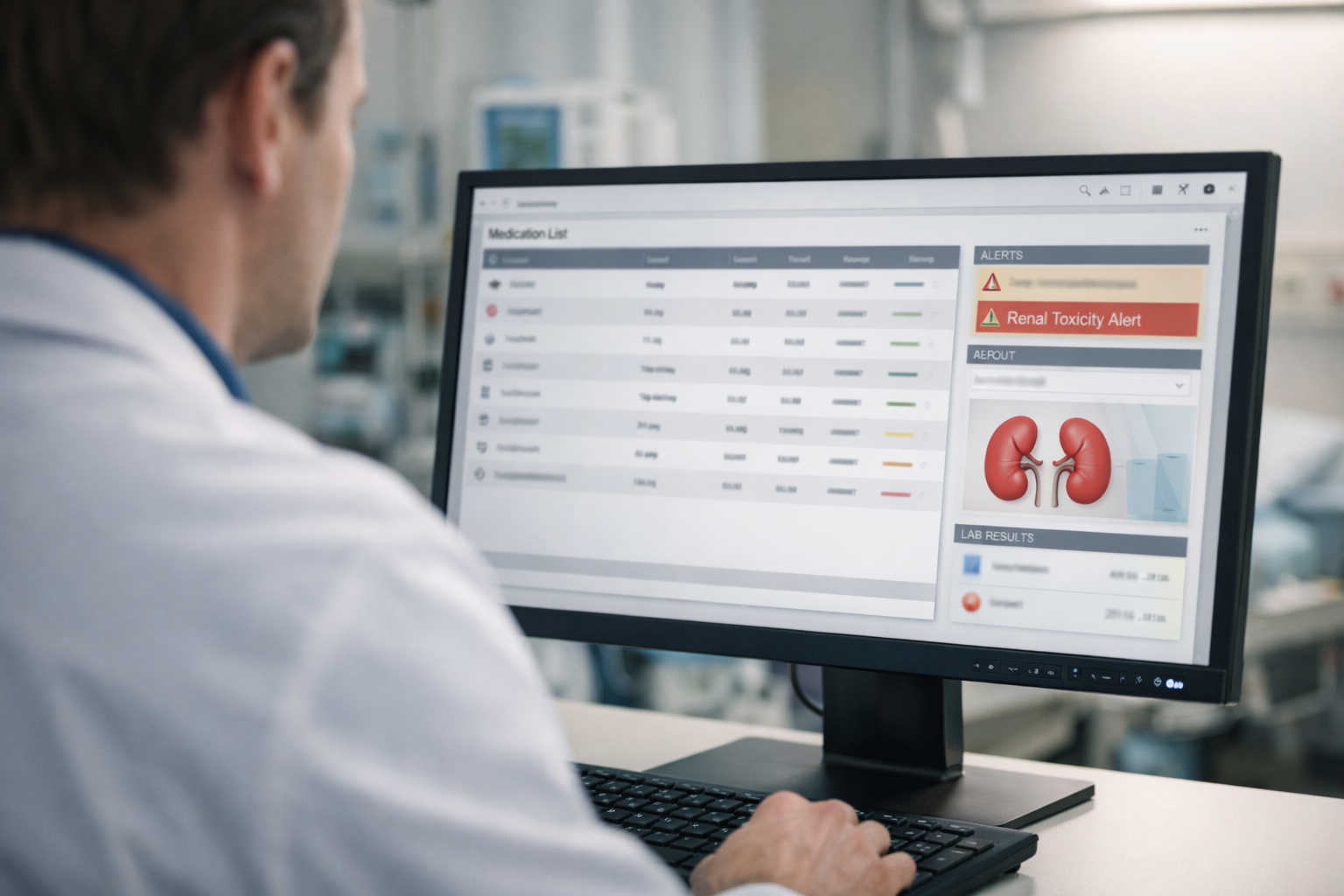
Riskbase provides information about a medication’s risk profile by highlighting the potential pharmacodynamic drug interactions to healthcare professionals. In addition to information on adverse reactions, Riskbase offers detailed guidance on how to manage and mitigate these risks. The information in Riskbase complements Inxbase’s drug interactions and is designed to minimise over-alerting along with Inxbase.
Key characteristics
of Riskbase
- Contains over 20 000 evaluations of drug risk profiles detailed through 11 different adverse effects
- Description of consequences and outcomes of each adverse effect
- Recommendations on avoiding, handling and monitoring adverse effects
- Inclusion of prescription and over-the-counter drugs

Detailed information on Riskbase
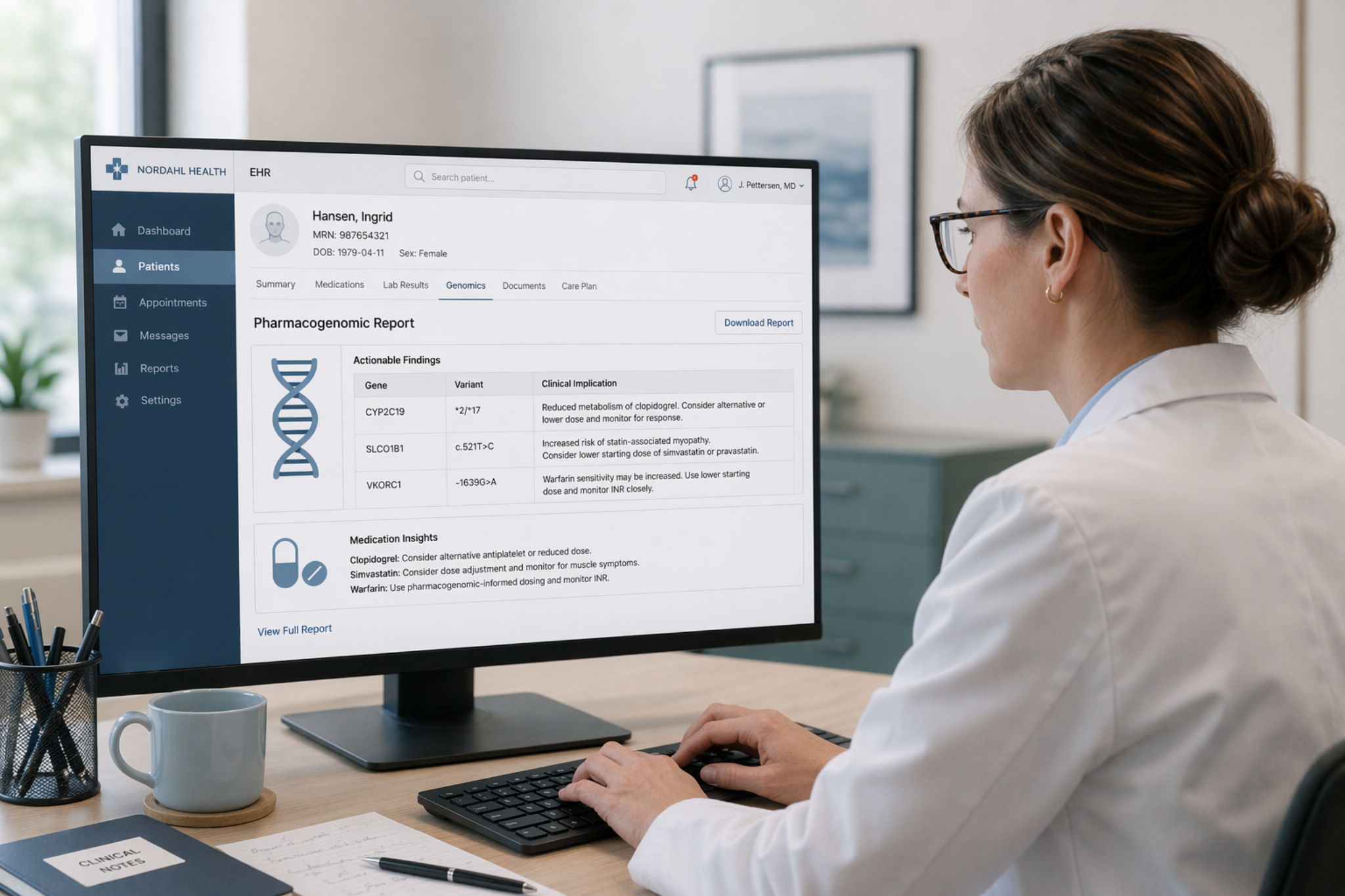
Drug interactions can be divided into pharmacokinetic interactions, which can lead to a change of exposure of one or several of the drugs involved, and pharmacodynamic interactions, which involve a change of the effects of the drug at the receptor or target organ, or adverse effects on other physiological systems. The drug interaction warning system Inxbase, which is based on individual drug pairs, is not suitable for creating warnings against all potential pharmacodynamic interactions. By creating an alert for every single drug pair, an overwhelming amount of alerting would ensue within electronic health record systems. Therefore, to minimise over-alerting, Riskbase is designed as a complementary database to Inxbase.
Adverse effects in Riskbase
Riskbase contains over 20,000 evaluations of the risk profiles of more than 1,800 drugs in relation to the following adverse effects:
- Anticholinergic effect
- Constipation
- Sedation
- Orthostatic effect
- Risk of bleeding
- Serotonergic effect
- Risk of seizures
- QT-prolongation
- Renal toxicity
- Potassium balance
- Sodium balance
Classification methodology
Each drug has been graded for the adverse effects included in Riskbase. Based on the total risk score of the medication, a classification is determined. Classifications and recommendations are presented using a system of traffic lights and letters ranging from A to D. In Riskbase, the D level does not signify an absolute contraindication but provides an indication that the overall risk is high.
| D | There is a high risk |
| C | There is a moderately increased risk |
| B | There is a somewhat increased risk |
| A | No known pharmacological or clinical basis for an increased risk |
Languages and local drug registries
Medbase content is available in English and more than ten additional languages. To further improve local usability, we accommodate national, local and customer-specific drug product registries, allowing access to information using local drug product names and IDs.
Integration with local electronic health record (EHR) systems is straightforward, enabling smooth and efficient use of regularly updated Medbase drug information across various countries.
Latest information from trusted sources
All information is based on scientific evidence. We refer to published, peer-reviewed research articles from PubMed, a reliable source of biomedical and life sciences literature, as well as regulatory authority approved documents, such as the Summaries of Product Characteristics (SPC) of drugs.
Medbase Knowledgebase is continuously updated to ensure the inclusion of the latest information.
Supporting informed decisions for safe drug use
Enhanced patient safety
Holistic approach to patient’s pharmacotherapy enabling individualised drug treatment.
100% Evidence-based
Fully referenced information with transparency to original sources: documents approved by regulatory authorities and peer-reviewed literature.
Clinical relevance
We provide comprehensive and concise information for safe use of drugs, accessible from one single source.
Digitalised doctor consultation
Designed to support busy clinical practice for safe use of drugs. All information is created and validated by physicians specialised in clinical pharmacology.
Newsroom
Discover our latest updates & customer references
- Hovstadius B, Hovstadius K, Astrand B, Petersson G. Increasing polypharmacy – an individual-based study of the Swedish population 2005-2008. BMC Clin Pharmacol. 2010 Dec 2;10:16.
- Davies EA, O’Mahony MS. Adverse drug reactions in special populations – the elderly. Br J Clin Pharmacol. 2015 Oct;80(4):796-807.
- Phansalkar S, van der Sijs H, Tucker AD, Desai AA, Bell DS, Teich JM, Middleton B, Bates DW. Drug-drug interactions that should be non-interruptive in order to reduce alert fatigue in electronic health records. J Am Med Inform Assoc. 2013 May 1;20(3):489-93.
- Ancker JS, Edwards A, Nosal S, Hauser D, Mauer E, Kaushal R. Effects of workload, work complexity, and repeated alerts on alert fatigue in a clinical decision support system. BMC Med Inform Decis Mak. 2017 Apr 10;17(1):36.
- Carollo M, Crisafulli S, Vitturi G, Besco M, Hinek D, Sartorio A, Tanara V, Spadacini G, Selleri M, Zanconato V, Fava C, Minuz P, Zamboni M, Trifirò G. Clinical impact of medication review and deprescribing in older inpatients: A systematic review and meta-analysis. J Am Geriatr Soc. 2024 Jun 1